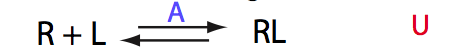
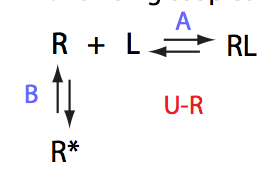
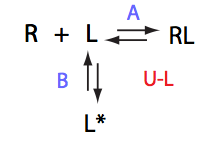
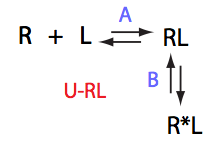
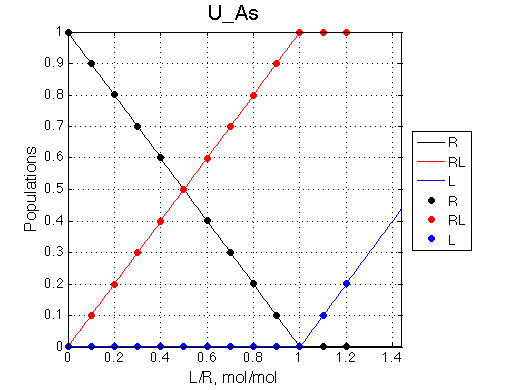
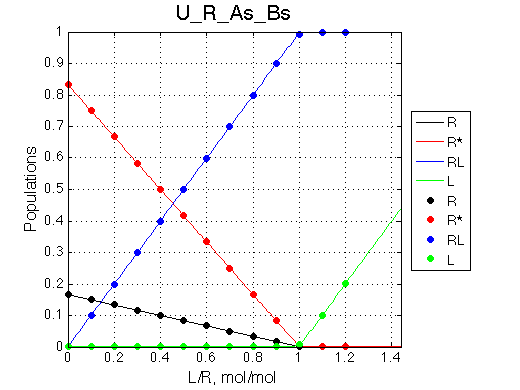
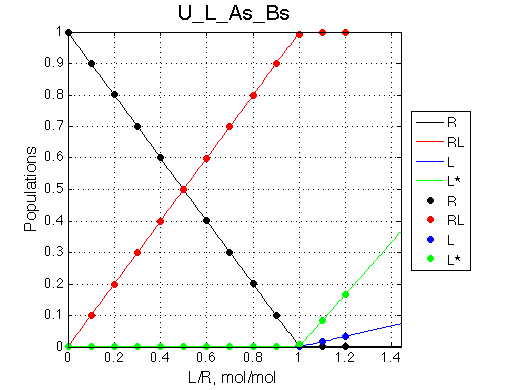
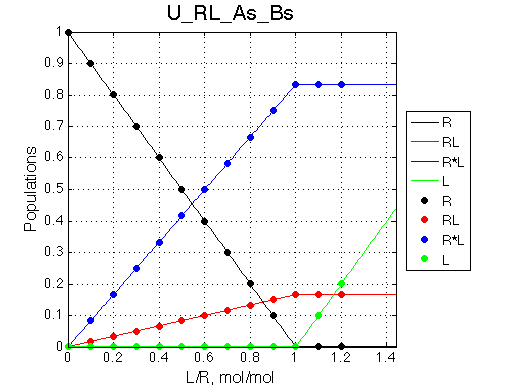
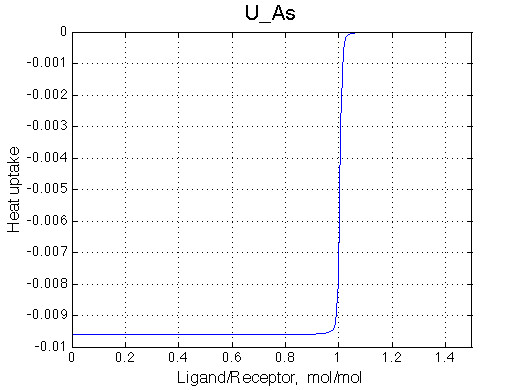
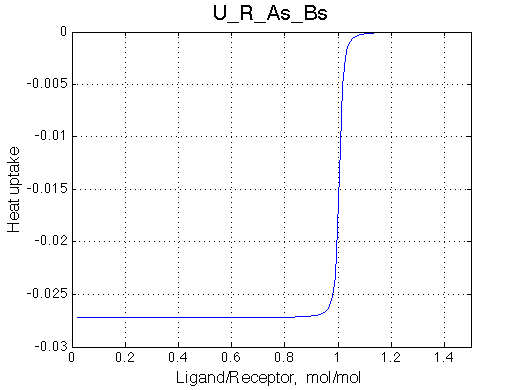
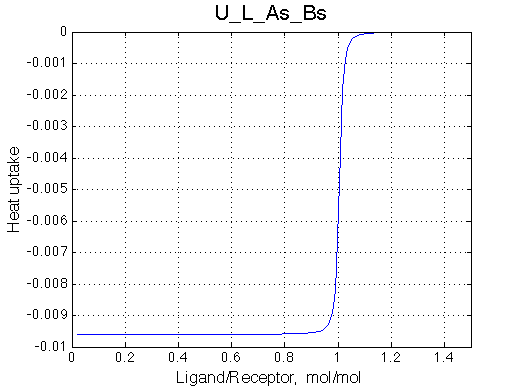
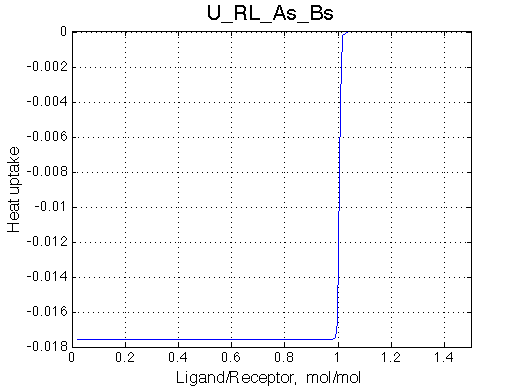
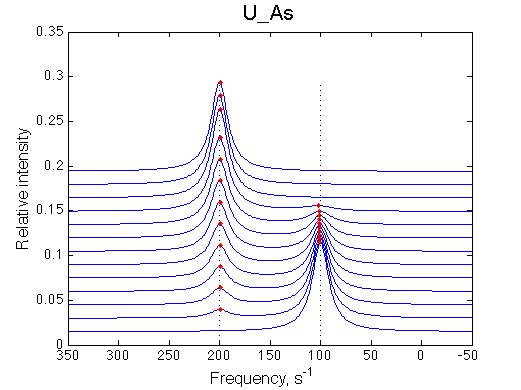
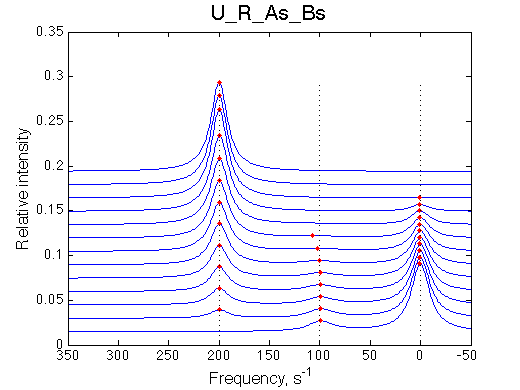
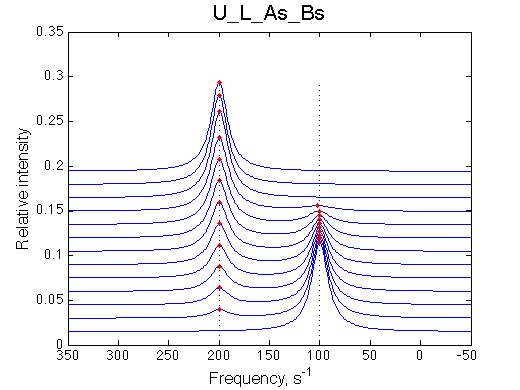
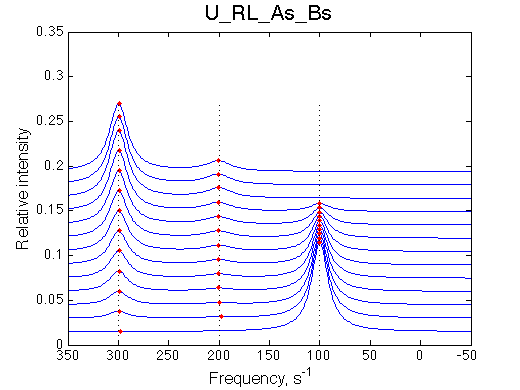
Here we will compare outcomes of the four alternative models for conditions when all transitions are very slow. I choose binding affinity of 108 M-1 and off-rate and isomerization constants of 1 s-1. R and RL are separated by 100 s-1 and R* or R*L - by 100 s-1 from corresponding species. Isomerization equilibrium is shifted 5:1 toward *-forms.
Location: As_Bs/
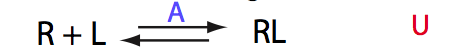 |
 |
 |
 |
| U_As.txt | U_R_As_Bs.txt | U_L_As_Bs.txt | U_RL_As_Bs.txt |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
Now we will imagine performing direct and reverse titrations and compare behaviors originating from different models.
When all reactions are very slow it is a key to observe and monitor minor peaks. Minor peaks may be coming from (1) true slow exchange between major and minor conformers, and (2) from minor population of the labeled component due to chemical modifications or other preparation artifacts.
If the mechanism is simple binding with no coupled isomerization we will observe the same type of spectral changes in both direct and reverse titrations because neither of the species has populations split between alternative conformers:
L -> R titration R -> L titration


(Here I simply duplicate image of U_As lineshapes for the reverse titration series. Real titration data will have different chemical shift separations and line widths associated with R and L creating different spectral appearance with the same pattern of disappearing and newly appearing peaks).
If the mechanism involves isomerization of one of the binding partners then direct and reverse titrations will give different complementary results represented by the U_R and U_L models:


The component that has slow interconversion should exhibit minor peaks disappearing with the titration progress and no new minor peaks should appear in the bound spectrum. Reverse titration, when isomerizing component is unlabeled is not affected by the conformational change in the ligand and displays two-state binding spectra.
Minor peaks in protein spectra may be artifacts of protein preparation procedure due to chemically modified groups in vicinity of binding surface. Therefore, when the system is in slow exchange it is important to verify whether the minor peaks represent the minor forms that are in exchange with the major form by using ZZ-exchange experiment (Farrow et al. 1994) though it may be challenging due to intrinsic low intensity of signals of the minor conformers.
If isomerization occurs in the RL complex then the direct and reverse titration with produce similar type of spectra where a single peak of the original species disappears and the two peaks of the bound form are detected. Isomerization of the complex most likely will perturb signals of both binding partners in the complex so both reverse and direct titration spectra will show this pattern.
L -> R titration R -> L titration


This is slow exchange system so we have to group titration points near equivalence to better describe curvature of the populations graph:
setup.txt
I will choose KB =20 to ensure the spectral appearance is indistinguishable from the one produced by the 2-site system. For the exchange rate constant to be equal to 1 s-1 in this case the reverse rate k2B has to be adjusted to 1/(20+1)=0.0476 s-1 .
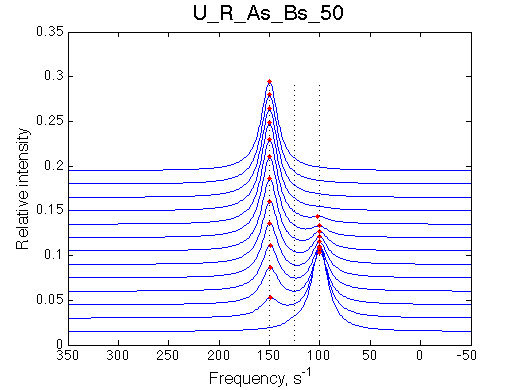
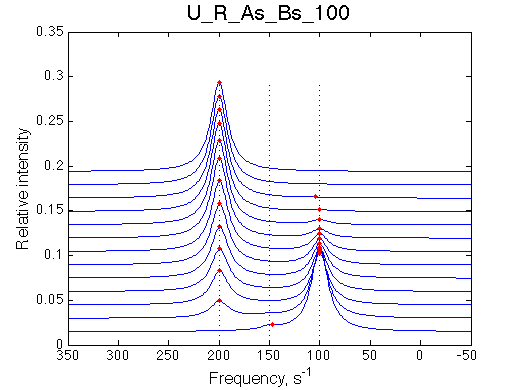
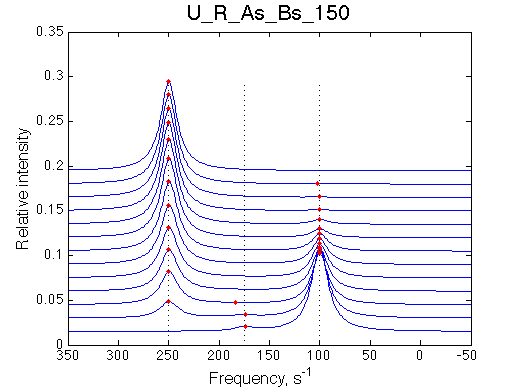
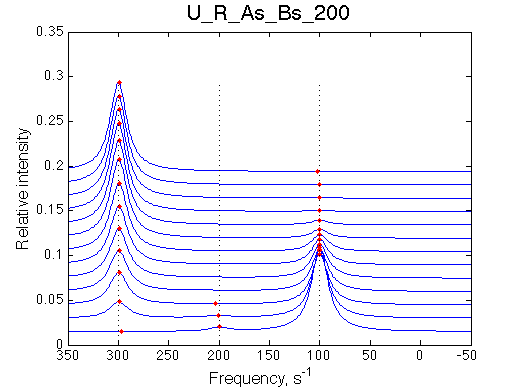
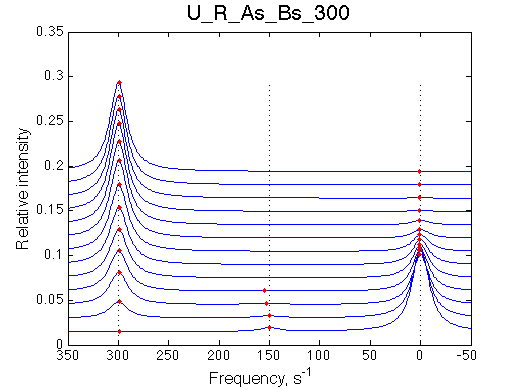
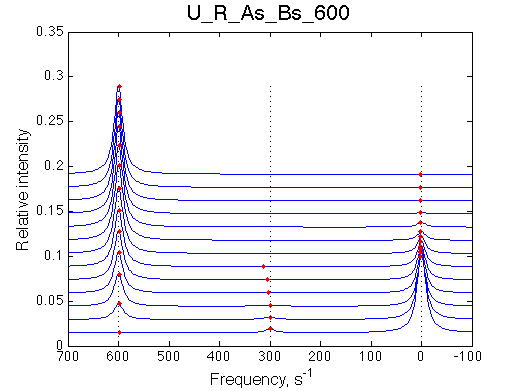
I will set full separation between end species to 50, 100, 150, 200 and 300 s-1 , while keeping the intermediate species in the middle.
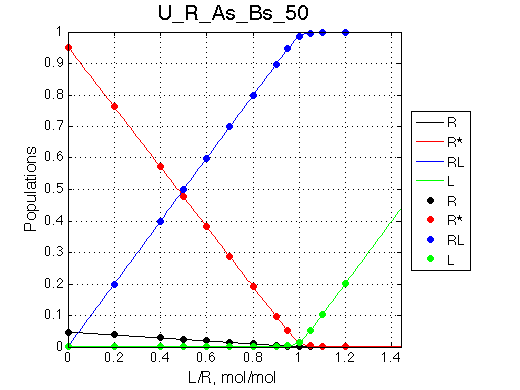
50 s-1 |
100 s-1 |
150 s-1 |
200 s-1 |
300 s-1 |
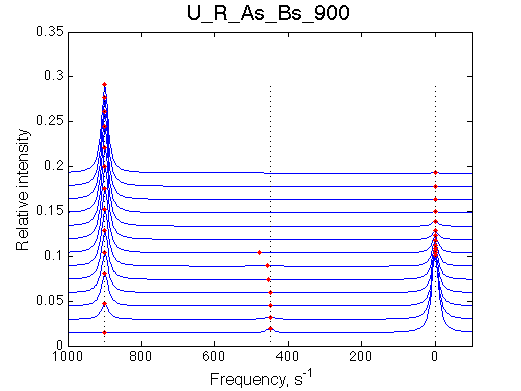
600 s-1 | 900 s-1 | |
| U_R | U_R_As_Bs_50 | U_R_As_Bs_100 | U_R_As_Bs_150 | U_R_As_Bs_200 | U_R_As_Bs_300 | Simulate setup2 U_R_As_Bs_600 | Simulate setup3 U_R_As_Bs_900 |
 |
 |
 |
 |
 |
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
|
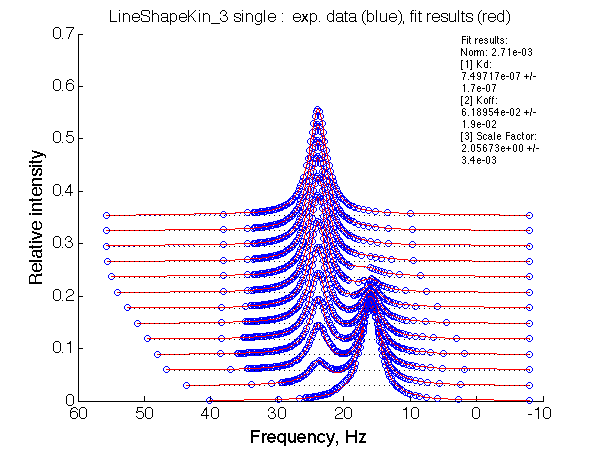
| LSQCURVFIT converged to a solution Determination of parameter uncertainties... Norm (sum of squared residuals): 2.71e-03 [1] Kd: 7.49717e-07 +/- 1.7e-07 [2] Koff: 6.18954e-02 +/- 1.9e-02 [3] Scale Factor: 2.05673e+00 +/- 3.4e-03 |
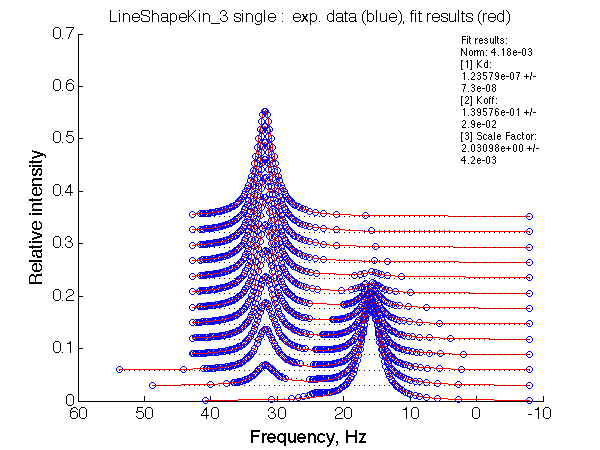
LSQCURVFIT converged to a solution Determination of parameter uncertainties... Norm (sum of squared residuals): 4.18e-03 [1] Kd: 1.23579e-07 +/- 7.3e-08 [2] Koff: 1.39576e-01 +/- 2.9e-02 [3] Scale Factor: 2.03098e+00 +/- 4.2e-03 |
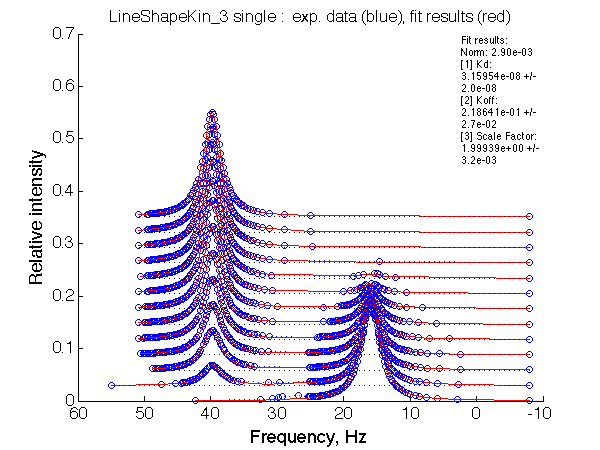
LSQCURVFIT converged to a solution Determination of parameter uncertainties... Norm (sum of squared residuals): 2.90e-03 [1] Kd: 3.15954e-08 +/- 2.0e-08 [2] Koff: 2.18641e-01 +/- 2.7e-02 [3] Scale Factor: 1.99939e+00 +/- 3.2e-03 |
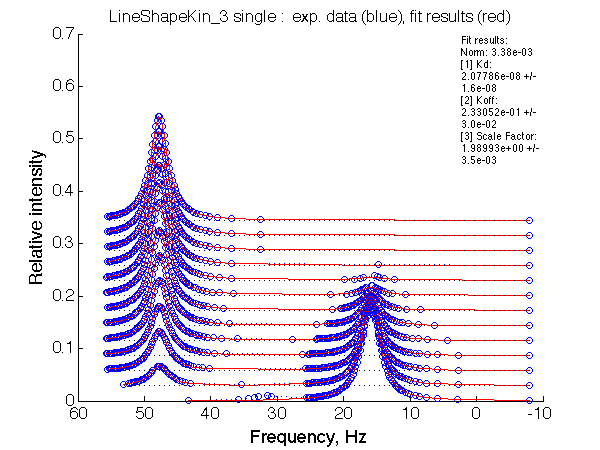
LSQCURVFIT converged to a solution Determination of parameter uncertainties... Norm (sum of squared residuals): 3.38e-03 [1] Kd: 2.07786e-08 +/- 1.6e-08 [2] Koff: 2.33052e-01 +/- 3.0e-02 [3] Scale Factor: 1.98993e+00 +/- 3.5e-03 |
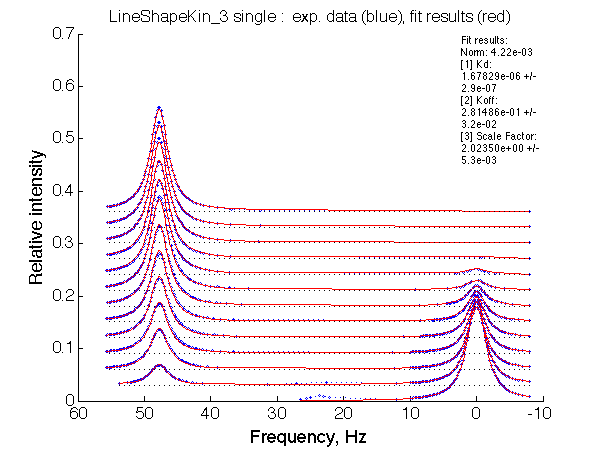
LSQCURVFIT converged to a solution Determination of parameter uncertainties... Norm (sum of squared residuals): 4.22e-03 [1] Kd: 1.67829e-06 +/- 2.9e-07 [2] Koff: 2.81486e-01 +/- 3.2e-02 [3] Scale Factor: 2.02350e+00 +/- 5.3e-03 |
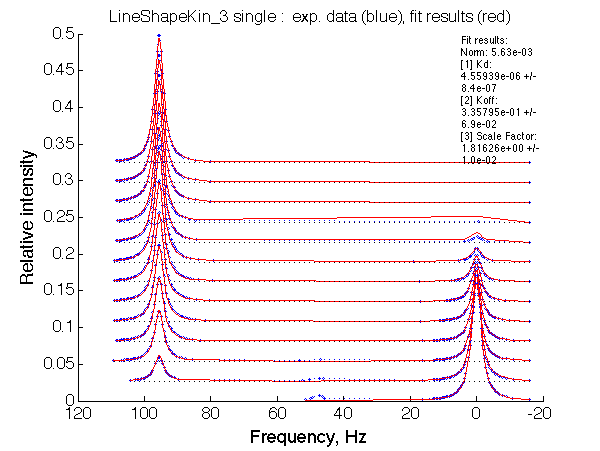
LSQCURVFIT converged to a solution Determination of parameter uncertainties... Norm (sum of squared residuals): 5.63e-03 [1] Kd: 4.55939e-06 +/- 8.4e-07 [2] Koff: 3.35795e-01 +/- 6.9e-02 [3] Scale Factor: 1.81626e+00 +/- 1.0e-02 |
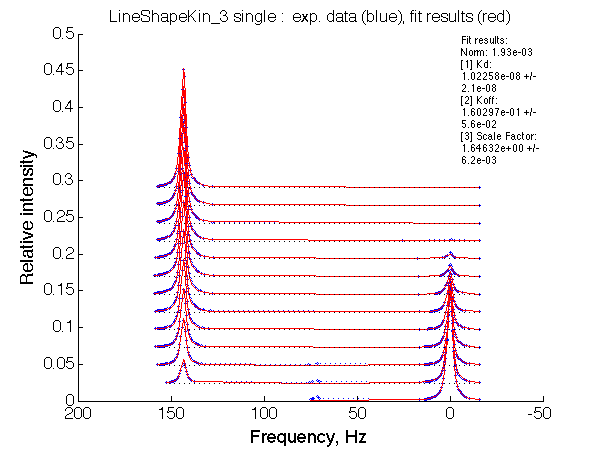
LSQCURVFIT converged to a solution Determination of parameter uncertainties... Norm (sum of squared residuals): 1.93e-03 [1] Kd: 1.02258e-08 +/- 2.1e-08 [2] Koff: 1.60297e-01 +/- 5.6e-02 [3] Scale Factor: 1.64632e+00 +/- 6.2e-03 |
No reliable dependence of the fitted parameters on chemical shift separation is detected.
Back to Analysis of Direct and Reverse Titrations